Updated Data for Indoximod Plus KEYTRUDA® (pembrolizumab) Demonstrate Improvement of Response Rate for Patients with Advanced Melanoma
Pivotal Trial of Indoximod in Advanced Melanoma to Include Both PD-1 Inhibitors, KEYTRUDA (pembrolizumab) and OPDIVO® (nivolumab)
This Smart News Release features multimedia. View the full release here: http://www.businesswire.com/news/home/20170907005441/en/
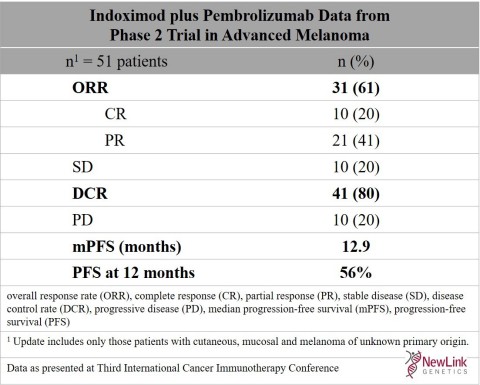
Indoximod plus pembrolizumab data from Phase 2 trial in advanced melanoma (Graphic: Business Wire)
The presentation entitled, "Combined Inhibition of the IDO and PD-1 Pathways Improves the Response Rate for Patients with Advanced Melanoma", showed an improvement over previously reported results presented at the AACR Annual Meeting 2017 for both the Complete Response rate (CR) and the Overall Response Rate (ORR) for patients1 who received indoximod plus pembrolizumab. Evaluable patients were defined as those having at least one on-treatment imaging study.
Key findings in the updated data reported today:
- Improvement in Complete Response (CR) to 20% (10/51 patients) compared to CR of 12% (6/51 patients)
- The Progression-Free Survival (PFS) by RECIST criteria was 56% at one year with median PFS (mPFS) of 12.9 months
"We are encouraged by the progression-free survival and the improvement
in complete responses observed in the trial," said
| Indoximod plus Pembrolizumab Data from Phase 2 Trial in Advanced Melanoma | |
| n1 = 51 patients | n (%) |
| ORR | 31 (61) |
| CR | 10 (20) |
| PR | 21 (41) |
| SD | 10 (20) |
| DCR | 41 (80) |
| PD | 10 (20) |
| mPFS (months) | 12.9 |
| PFS at 12 months | 56% |
|
overall response rate (ORR), complete response (CR), partial
response (PR), stable disease (SD), disease control |
|
| 1 Update includes only those patients with cutaneous, mucosal and melanoma of unknown primary origin. | |
|
Data as presented at |
|
Indoximod in combination with pembrolizumab was well-tolerated. The most common all-grade adverse events were fatigue, headache, and nausea. Three patients experienced grade 3 serious adverse events (SAE) possibly attributed to indoximod. Three patients experienced SAEs that led to discontinuation of treatment. There were no treatment related deaths.
Pivotal Trial of Indoximod in Advanced Melanoma to Include Both PD-1 Inhibitors, KEYTRUDA (pembrolizumab) and OPDIVO (nivolumab)
The pivotal trial has been designed as a large-scale (600 patients) trial in Stage III unresectable and metastatic stage IV melanoma. The trial will have a one to one randomization between indoximod plus KEYTRUDA (pembrolizumab) or OPDIVO (nivolumab) compared to single agent PD-1 inhibitor. The co-primary endpoints of the study are PFS by RECIST criteria and Overall Survival (OS).
"Our team is excited to move forward with this pivotal trial," said
About Indoximod
Indoximod is an investigational, orally available small molecule targeting the IDO pathway. The IDO pathway is one of the key immuno-oncology targets involved in regulating the tumor microenvironment and immune escape.
About
KEYTRUDA® is a registered trademark of
OPDIVO® is a registered trademark of Bristol-Myers Squibb Company.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements of
View source version on businesswire.com: http://www.businesswire.com/news/home/20170907005441/en/
Investor Contact:
Director
of Investor Relations
lmiller@linkp.com
or
Media:
LaVoieHealthScience
AVP, Public & Media Relations
amastrangelo@lavoiehealthscience.com
Source:
News Provided by Acquire Media

