NewLink Genetics Describes the Differentiated Mechanism of Action of Indoximod in AACR Poster Presentation
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20180417005885/en/
(Graphic: Business Wire)
“The data demonstrate that indoximod has a unique mechanism of action,
remarkably differentiated from IDO enzymatic inhibitors. This different
mechanism may contribute to antitumor immune responses in the IDO
pathway and through activity independent of IDO,” said
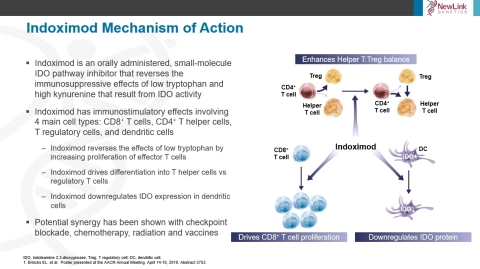
The data suggest that indoximod regulates the differentiation of helper T cells toward an immuno-stimulatory helper function and downregulates genes that control the differentiation of T cells into immuno-suppressive regulatory T cells (Tregs) in an AhR dependent manner. This leads to an increase in the ratio of helper T cells to Tregs. Additionally, it was shown that indoximod reduces the level of IDO protein in dendritic cells in vitro, leading to increased stimulation of CD8 T cell proliferation and reduced production of kynurenine. Moreover, indoximod stimulation of mTOR in T cells appears to increase the proliferation of effector T cells in an IDO and TDO-independent manner. Through this mechanism, indoximod may be able overcome the effects of Trp degradation mediated by both IDO and TDO. Thus, in addition to opposing immunosuppression mediated by the IDO pathway, indoximod may drive antitumor immune responses independent from IDO.
In summary, indoximod has immunostimulatory effects involving 4 main cell types: CD8+ T cells, T helper cells, T regulatory cells, and dendritic cells. Indoximod appears to function through three main mechanisms to inhibit the IDO pathway:
- Reversing the effects of low tryptophan by increasing proliferation of effector T cells
- Increasing the ratio of T helper to T regulatory cells by both favoring differentiation of activated CD4 T cells into helper T cells and directly reprogramming T regulatory cells into helper T cells
- Downregulating IDO expression in dendritic cells
About Indoximod
Indoximod is an investigational, orally available small molecule targeting the IDO pathway. The IDO pathway is a key immuno-oncology target involved in regulating the tumor microenvironment and immune escape. Indoximod is being evaluated in combination with treatment regimens including anti-PD-1/PD-L1 agents, cancer vaccines, radiation and chemotherapy across solid and liquid tumors.
About NewLink Genetics Corporation
NewLink Genetics is a late-stage biopharmaceutical company focusing on
discovering, developing and commercializing novel immuno-oncology
product candidates to improve the lives of patients with cancer.
Editor’s Note: Please see infographic on Indoximod Mechanism of Action
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements of
View source version on businesswire.com: https://www.businesswire.com/news/home/20180417005885/en/
Source:
Investor:
NewLink Genetics
Lisa Miller, 515-598-2555
Director
of Investor Relations
lmiller@linkp.com
or
Media:
LaVoieHealthScience
Sharon
Correia, 617-374-8800, ext. 105
VP, Integrated Communications
scorreia@lavoiehealthscience.com

